Rubber ranks among the most versatile materials on the planet. Its fame stems from the fact that engineers can transform it into anything from an incredibly soft, stretchy band to a solid bowling ball. Categorize rubber into two main types one is Natural and Synthetic.
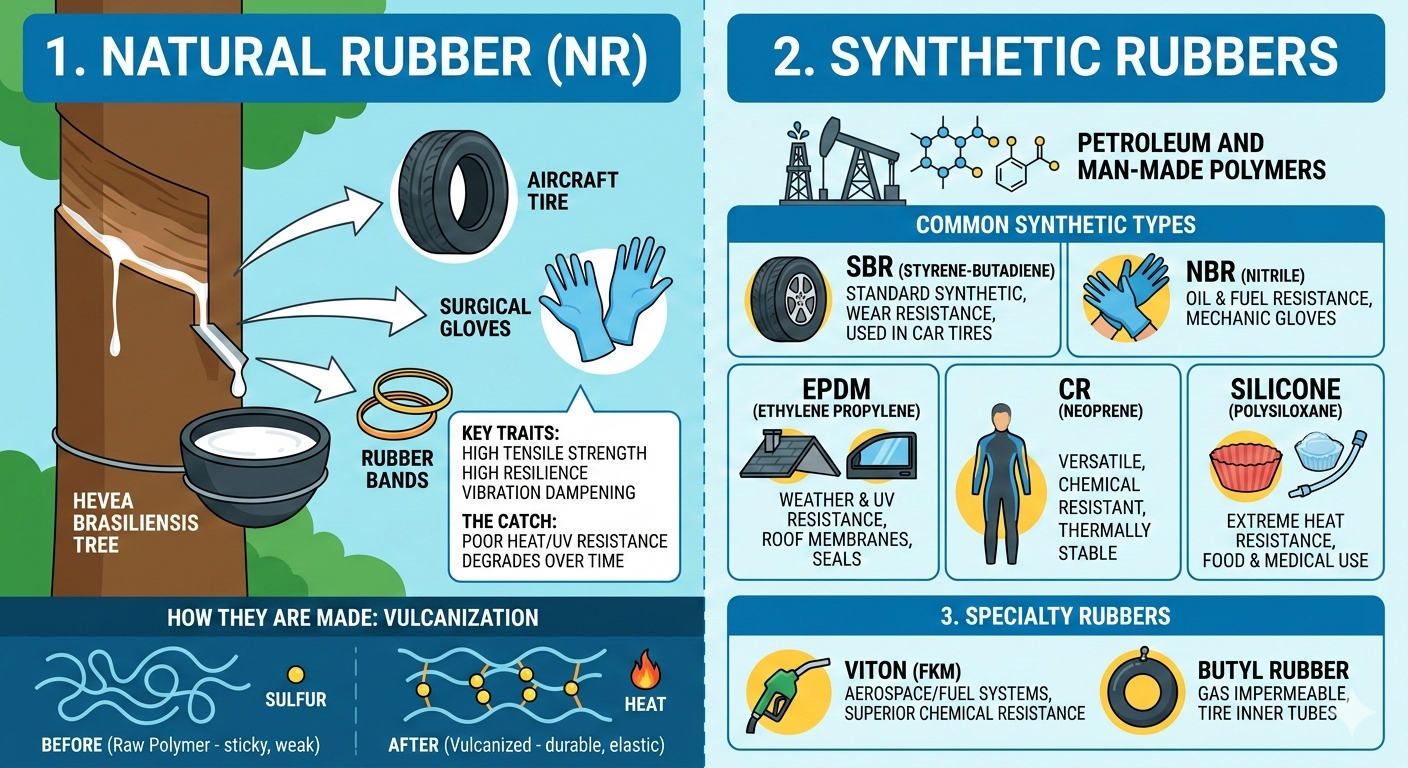
1. Natural Rubber (NR)
Derived from the latex sap of the Hevea brasiliensis tree, natural rubber is the “OG” material.
“Nevertheless, even with all our modern technology, scientists still haven’t perfectly replicated its unique properties.”
- Key Traits: Exceptional tensile strength, high resilience (snap-back), and vibration dampening.
- Best For: Tires (especially heavy-duty aircraft and truck tires), surgical gloves, and rubber bands.
- The Catch: It doesn’t handle heat, oil, or UV light very well—it tends to degrade and get brittle over time when exposed to the elements.
2. Synthetic Rubbers
Synthetic rubbers are man-made polymers derived from petroleum. Engineers “tune” these rubbers to handle specific environments where natural rubber would fail.
Common Synthetic Types:
| Type | Common Name | Best Known For… |
| SBR | Styrene-Butadiene | The “standard” synthetic. Used in 50% of car tires for its wear resistance. |
| NBR | Nitrile | Oil and fuel resistance. This is what “Blue” mechanic gloves are made of. |
| EPDM | Ethylene Propylene | Weather resistance. Used for roof membranes and car window seals. |
| CR | Neoprene | Versatility. Chemical resistant and thermally stable. Think wetsuits and laptop sleeves. |
| Silicone | Polysiloxane | Heat resistance. Safe for food and medical use. Can handle extreme cold and heat. |
3. Specialty Rubbers
- Viton (FKM): The “Gold Standard” for chemical resistance. It’s used in aerospace and automotive fuel systems because it survives temperatures that would melt other rubbers.
- Butyl Rubber: Unique because it is gas impermeable. If you have an inner tube or a tubeless tire that holds air for months, thank butyl rubber.
How they Created?
Generally speaking, most rubber—whether natural or synthetic—must undergo a specific process called Vulcanization. Without this crucial step, the material remains highly unstable; for instance, it becomes sticky when hot and brittle when cold. In contrast, by adding sulfur and heat, we trigger a vital chemical reaction. Consequently, these elements create cross-links between the polymer chains, thereby transforming the raw latex into the durable, bouncy material we recognize today. Ultimately, this transformation ensures the rubber can withstand extreme conditions.